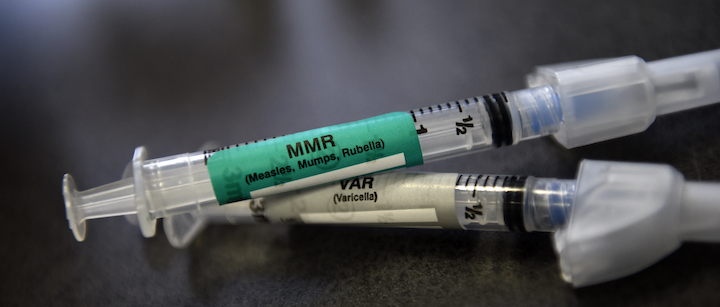
Unpacking the CDC’s Recent Shift on the MMRV Vaccine: What’s True, What’s Misleading
Recently, the Centers for Disease Control and Prevention (CDC) announced a pivotal change in its vaccination guidelines concerning the combined MMRV (measles, mumps, rubella, and varicella) vaccine. On September 18, the CDC’s Advisory Committee on Immunization Practices (ACIP) voted to de-emphasize the use of the MMRV vaccine as the preferred option for children under 4 years old, favoring separate MMR (measles, mumps, rubella) and chickenpox vaccines instead. The primary reasoning provided was a slightly increased risk of febrile seizures, a benign seizure related to fever, associated with the combined vaccine in young children.
Assessing the Safety Concerns and the Evidence Base
The focus of safety discussions centers on febrile seizures, which are temporary, typically harmless seizures triggered by fever, occurring most often between 14 and 18 months of age. The CDC’s own data shows that children receiving the MMRV vaccine as their first dose experience approximately one additional febrile seizure per 2,300 to 2,600 children compared to those receiving separate MMR and varicella vaccines. This figure stems from extensive safety monitoring, including CDC’s Vaccine Safety Datalink and multiple clinical studies, which have consistently demonstrated that febrile seizures, though frightening, do not cause long-term health issues such as neurological damage or cognitive problems.
Leading experts like Dr. H. Cody Meissner, a pediatric infectious disease specialist, have affirmed that these seizures are “rarely associated with long-term problems” and “do not impair neurocognitive development” — an assessment corroborated by numerous peer-reviewed studies. This conclusion aligns with the broader medical consensus that the vaccines are safe, with the slight increased risk of febrile seizures being a manageable and transient side effect.
The Decision-Making Process: Was It Proper?
However, questions have been raised regarding the process through which ACIP arrived at this new recommendation. Critics, including former CDC officials and public health commentators, argue that the process was expedited and lacked the comprehensive evidence review normally required for such policy shifts. Unlike standard procedures that involve systematic assessments over months or even years, this recommendation was made on a compressed schedule, with some experts claiming that no significant new safety data justified the change.
- CDC’s traditional process involves detailed reviews of scientific evidence, feasibility, and public input; yet, sources indicate that in this case, the preparation time was notably shorter than customary.
- Major stakeholders, including the American Academy of Pediatrics, did not participate directly in the decision, suggesting potential gaps in collaborative review.
- Critics emphasize that the abrupt change could influence vaccine coverage and parental choice—parameters vital to maintaining herd immunity.
Moreover, internal dissent within the CDC has been publicly aired, with former officials expressing frustration over politicization and process shortcuts. Dr. Demetre Daskalakis, who resigned citing concerns over CDC policy-making, highlighted that this fast-tracked decision diverges from established evidence-based protocols.
Implications for Public Confidence and Vaccine Uptake
The practical consequence of removing the MMRV vaccine as a default option could lead to reduced vaccine adherence or delayed immunizations, especially if parents perceive the separate injections as less convenient or less effective. Data suggests that around 15% of children currently receive MMRV as their initial vaccine, with this figure varying across states. Any decline in vaccination rates risks increasing susceptibility to outbreaks of measles, mumps, and chickenpox, conditions which vaccination has dramatically suppressed over past decades.
As safety data remains robust, the primary issues lie in perceptions. Critics warn that such policy shifts—especially if perceived as driven by political or bureaucratic motives rather than scientific necessity—may erode public trust. Maintaining transparent, thorough decision-making processes is essential to ensure community confidence in childhood immunizations, which are pillars of public health and democracy.
Conclusion
In an age where misinformation can spread as rapidly as viruses, transparency and adherence to scientific rigor are vital. The CDC’s decision to #change its vaccine recommendations should be scrutinized not just for its immediate safety rationale but also for its process integrity. Responsible, science-based policymaking fortifies public trust, which is indispensable to the success of vaccination programs and the health of future generations. Ensuring that health authorities remain committed to transparent, evidence-driven decisions upholds the democratic ideal that true safety and choice are fundamental rights for all parents and children.